Abstract
Introduction: GATA2 haploinsufficiency syndrome (G2HS) is an autosomal dominant germline predisposition state with broad phenotypic variability and high risk of developing myeloid neoplasms (74.3%, median age of onset 17 years; Homan CC, et al. 2021). Mechanisms resulting in myeloid transformation remain to be elucidated. To identify molecular changes and relevant drivers of gene expression prior to progression, we performed serial, single cell multiomics on a proband with G2HS and ASXL1 mutant clonal hematopoiesis (CH) over 7 years.
Case report: The proband was diagnosed with G2HS at age 19 years when he presented with recurrent infections, scrotal and penile lymphedema and cytopenias. Bone marrow (BM) biopsy at diagnosis was hypocellular with minimal atypia. Next generation sequencing (NGS) identified a novel pathogenic variant in GATA2 (NM_001145661.2: c.1143+200_1198delinsC; Dorn J et al, 2017) confirmed to be germline, and a somatic pathogenic variant in ASXL1 (NM_015338.6: c.2638dupA; p.Thr880Asnfs*2) with a variant allele frequency (VAF) of 11%, indicative of CH.
Methods: After IRB approval, clinical data was abstracted from the medical records. Four aliquots of BM mononuclear cells from BM biopsies obtained 6 months apart were subjected to targeted NGS, single cell RNA (scRNA-seq) and scATAC-seq. Preprocessing and poor-quality cell filtering were performed using the Cell Ranger pipeline. For scRNA-seq, filtered counts were clustered and annotated using Seurat v4.1.0 onto a CITEseq PBMC reference (Hao Y, et al. 2021). Genes were ranked within each cluster between samples based on their average log-fold change, and gene set enrichment analysis (GSEA) was performed using the fgsea package v1.16.0. In parallel, filtered scATAC-seq data was integrated with scRNA using the cluster-label transfer from Signa v1.5.0 and Seurat v4.1.0. Using MAESTRO (Wang C, et al. 2020), we predicted the transcriptional factors (TF) regulating the expression from differentially expressed genes and differential peaks between samples based on the enrichment scores from algorithms LISA (Qin Q, et al. 2020) and GIGGLE (Layer RM, et al. 2018) for RNAseq and ATACseq respectively.
Results: Serial blood cell counts, blood indices and BM biopsies over 7 years remained stable (macrocytosis, monocytopenia and neutropenia), with the BM demonstrating stable hypocellularity (10-20%) with minimal atypia and with no evidence for progression to myelodysplastic syndrome (MDS). ASXL1 mutant VAF also remained stable (11 to 16% at last measurement). Quantitative flow cytometric assessments in blood revealed decreases in B and NK cells. Analysis of scRNA/scATAC-seq successfully identified monocytes, B-cells, CD4, CD8 and NK Cells yielding comparable counts to those obtained by flow cytometry.
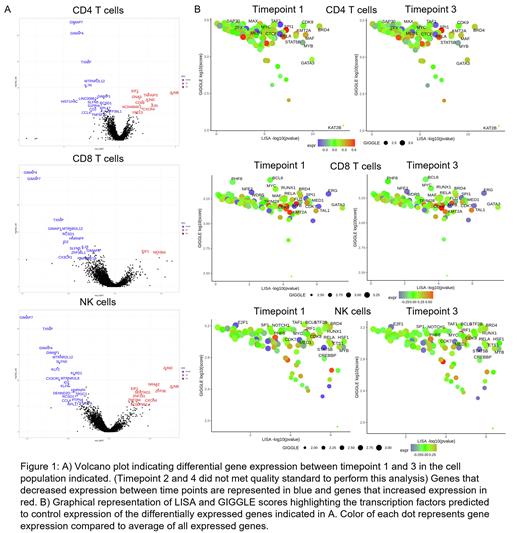
The G2HS-associated cytopenias resulted in very small numbers of monocytes and B cells being identified, and hence differential gene expression analysis was limited to CD4+, CD8+ T cells and NK cells, between time points 1 and 3 (Figure 1A). Differentially expressed genes showed aberrant enrichment of downregulated genes in pathways related to hematopoietic stem cell (HSC) maturation in all three populations, while upregulated genes were enriched in cell cycle and innate immune responses. Analysis of regulatory drivers identified BRD4 as the main TF controlling the differentially expressed genes in CD4+ T cells and NK cells, and ERG in CD8+ T cells. The expression of these TF did not change between timepoints. (Figure 1B)
Discussion: We present a GATA2 haploinsufficient proband who has remained stable and MDS-free over 7 years, even with ASXL1 mutant CH. Despite the stability in his blood counts and BM, single cell analysis has identified progressive qualitative changes impacting relevant pathways including HSC maturation, cell cycle regulation and innate immune responses in T and NK cells. In addition, our scATAC-seq data predicts BRD4 and ERG to be important epigenetic readers/TF that regulate these changes. We plan to study larger cohorts of patients and additional time points to understand transcriptional and epigenetic changes induced by GATA2 haploinsufficiency and elucidate the role of ASXL1 mutant CH in disease progression.
Disclosures
Abraham:Beckman Coulter: Honoraria; Enzyvant: Consultancy, Honoraria. Mangaonkar:Bristol Myers Squibb: Research Funding. Patnaik:Kura Oncology, Stem Line Pharmaceuticals: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal